Treating Mild to Moderate Asthma in Adolescence and Adults
The objectives for treating moderate, persistent asthma in adolescents and adults are to reduce symptoms, avert flare-ups, and restore normal lung function, to enhance the quality of life. For first-time patients, the initial therapy is tailored based on the severity of their condition. National guidelines suggest using either a single treatment or various combinations of inhaler therapies, each with a distinct mechanism of action (MOA), tailored to the disease’s progression. A stepwise treatment strategy starts with inhaled rescue medications for immediate symptom relief, followed by controller medications to prevent asthma attacks and manage chronic symptoms. Depending on the patient’s response, doses can be modified, or different combinations introduced.
Current Asthma Treatment Landscape
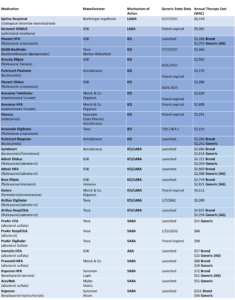
- Inhaled Corticosteroids (ICS) are one of the pillars of asthma therapy, treating the root cause of airway inflammation. While guidelines endorse ICS as the first-line controller therapy for children with persistent asthma, in adolescents and adults, they’re frequently paired with other inhaled medications for comprehensive symptom relief and control. ICS inhalers come in low, moderate and high-dose formulations, allowing for tailored dosing based on the severity of symptoms and the patient’s age.
- ICS/Long-Acting Beta-Agonists (LABA) Combination Inhalers stand as another cornerstone in asthma management. A LABA is a bronchodilator that, when paired with an ICS, functions both as a rescue and a controller. The bronchodilator widens the airways, enhancing the delivery and efficiency of the ICS in reducing lung inflammation. Historically, this class of inhalers saw competition among branded drugs, spearheaded by manufacturers like GSK, Astra Zeneca and Merck. However, the landscape shifted with the introduction of the first AB-rated generic equivalent to GSK’s Advair Diskus in 2019. Subsequent releases of other Advair Diskus generic alternatives further reduced prices. By July 2023, the market welcomed its first AB-rated generic counterpart to Astra Zeneca’s Symbicort as well.
- Short-Acting Beta2 Agonists (SABA) are bronchodilators designed to offer fast symptom relief from asthma. However, they don’t address the root inflammation causing the airway issues. Typically used as rescue inhalers, they’re administered as needed and can also be combined with controller inhalers for preventative measures. Emerging research suggests that simultaneous use of ICS and SABAs might offer advantages compared to the traditional pairing of formoterol (a LABA) and ICS for initial symptom relief. Previously, patients required two separate inhalers for the SABA/ICS mix, but recent advancements have introduced a combined SABA/ICS inhaler option. In January 2023, AstraZeneca introduced Airsupra, a combination of albuterol and budesonide. It stands as the first-in-class ICS/SABA combination approved for as-needed treatment or prevention of bronchoconstriction, as well as minimizing exacerbation risks in asthma patients aged 18 and older. While this solution offers convenience, generic versions of budesonide and albuterol inhalers purchased separately, are more cost effective. The retail price for Airsupra is set at $475 per inhaler, translating to an annual expense of $5,700.
Alternative Treatment Options
Alternate options to treat moderate persistent asthma include inhaled ICS with other agents such as a Long-Acting Muscarinic Antagonist (LAMA), a leukotriene modifier or slow-release theophylline. If dual therapy doesn’t achieve full symptom control, a third agent can be added.
Guidelines recommend triple therapy with the addition of a LAMA to ICS/LABA inhaler therapy if asthma is not controlled with the dual therapy combination. Recent studies indicate that incorporating a LAMA with the ICS/LABA combination decreases the yearly rate of severe exacerbations, with minor variation in severe side effects between the dual and triple therapies. However, triple therapy was associated with an increased risk of dry mouth and dysphoria. Currently, Trilegy Ellipta stands as the sole approved ICS/LAMA/LABA triple therapy product to treat asthma, carrying an annual price tag of $7,662.
See Appendix for a breakdown of therapies, their mechanisms of action, launch dates and their annual cost.
Management Considerations
The realm of asthma inhaler therapy has been historically competitive. Numerous treatment classes exist, with each housing multiple inhalers that offer comparable efficacy. While these inhalers might have varying dosage frequencies and device technologies, competitive pricing and rebates have traditionally shaped formulary management strategies in this sector. Yet, the emergence of several generic alternatives is reshaping these strategies.
Many brand-name products now offer authorized generics available at a slight discount to their branded counterparts. In some scenarios, prioritizing brand over generic might be financially sensible, especially if deals with brand manufacturers result in the lowest net costs. However, some manufacturers are discontinuing contracts for certain branded inhalers. This trend hints at a potential shift where generic competition might significantly reduce the cost of certain asthma inhalers.
Asthma, a pervasive and chronic lung condition, touches the lives of millions, necessitating comprehensive and effective treatment modalities. The rise of generic alternatives, coupled with advancements in inhaler technology, signifies an era of enhanced affordability and accessibility in asthma care. As we move forward, it’s vital for stakeholders to remain vigilant, understanding the importance of evolving treatments and the balance between efficacy and affordability in shaping a better future for asthma patients.
Appendix A: